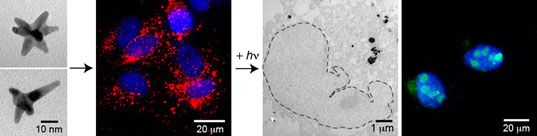
Direct visualization of interactions between drug-loaded nanoparticles and the cancer cell nucleus is crucial for understanding the mechanism behind cancer cell death. Nanoconstructs composed of nucleolin-specific aptamers and gold nanostars were actively transported to the nucleus and were found to induce major changes in nuclear phenotype: near the site of the nanoconstruct, nuclear envelope invaginations occurred. We have shown that the number of local deformations could be increased by ultra-fast, light-triggered release of the aptamers from the surface of the gold nanostars. Cancer cells with more nuclear envelope folding showed increased apoptosis as well as decreased cell viability. This newly revealed correlation between drug-induced changes in nuclear phenotype and increased therapeutic efficacy could provide new insight for localized cancer therapy.